Arrangement Of Electrons In An Atom Determine
In atomic physics and quantum chemistry the electron configuration is the distribution of electrons of an atom or molecule or other physical structure in atomic or molecular orbitals. Formula 2n 2 helps in the determination of the maximum number of electrons present in an orbit here n orbit number.

The Atom Chemistry Is My Jam Planetary Model Electron Configuration Chemistry Classroom
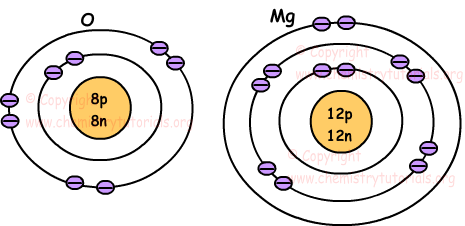
The electron arrangement of sodium 281 shows that sodium Na.

Arrangement of electrons in an atom determine. An electron arrangement is the way. For example a lithium atom Z3 A7 amu contains three protons found from Z three electrons as the number of protons is equal to the number of electrons in an atom and four neutrons 7 3 4. Is in group 1.
The specific arrangement of electrons in orbitals of an atom determines many of the chemical properties of that atom. Helps determine the number of electrons in the outermost energy level valence electrons. Electrons further away from the nucleus will have higher energy.
The electron arrangement of an atom can be worked out from its atomic number. So the second electron could go into the 1s orbital with the opposite spin of the first electron or it could go into the next orbital. Now as you can see in the third shell of the.
Is in period 3. Lithium has 3 protons and 3 neutrons and three electrons as well. Electrons closest to the nucleus will have the lowest energy.
Elements in this group are unreactive. This allows us to determine which orbitals are occupied by electrons in each atom. In addition the total number of electron shells an atom determines which period it belongs to.
What does the number and arrangement of electrons in. No two electrons in the same atom can have the same set of four quantum numbers Hunds Rule orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron and all electrons in singly occupied orbitals must have the same spin. The electron configuration is the arrangement of electrons in an atom.
So we have three ways to represent the electron arrangement in an atom. Total number of electrons in all shells. Each element emits a unique collection of lines.
Electrons that are in the first energy level energy level 1 are closest to the nucleus and will have the lowest energy. For example the atomic number of sodium is 11. For example the electron configuration of the neon atom is 1s2 2s2 2p6.
The orbital diagram the electron configuration and the energy diagram. The electron arrangement of an atom can be worked out from its atomic number. The chemical inactivity of noble gas atoms reflects the stable arrangement of their electrons one which other atoms cannot easily disturb.
Arrangement of electrons in atoms shells and subshells. Has only two possible values 12 -12. No the valance electrons determine the charge the atomic number is determined by the number of protons.
The electron arrangement also provides information about the number of valence electrons. All three ways are useful. The chemical behavior of an element can be understood by looking at the electron assignment of an atom of the element as compared to the electron assignments of neighboring noble gas atoms.
If two or more orbitals have the same energy. Ground-state electron configuration is the energy arrangement of the electrons for each element. The way in which the electrons are distributed in the shells of an atom is called the electron arrangement of the atom.
Describes arrangement of electrons in. Electrons can determine if an atom is a positive ions more protons than electrons or vice versa known as negative ions chemical reactions as atoms can donate or steal electrons and valency when it has 8 electrons in their shell known as Noble gases. Viewed simply electrons are arranged in shells around an atoms nucleus.
Each orbital can hold only two electrons which must be of opposite spin. Given an atomic number Z and mass number A you can find the number of protons neutrons and electrons in a neutral atom. 1travel in successively larger orbits and when an electron jumps from an outer orbit to an inner one it emits light.
For example the atomic number of sodium is 11. The distribution of electrons in an atom is called as Electronic Configuration. Rules for Electron Configuration.
What is the arrangement of electrons. No two electrons in the same atom can have the same set of four quantum numbers Hunds Rule orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron and all electrons in singly occupied orbitals must have the same spin. The atomic spectrum can be used to determine the composition of a material since it is different for each element of the periodic table.
Here electrons are arranged in energy levels or shells around the nucleus of an atom. An atoms electron shell can accommodate 2n. The valence electrons are the electrons in the highest energy level and the ones involved in ion and bond formation.
Take Sodium and Chlorine for example. Sodium has a electronic configuration of 281 and Chlorine has an electronic configuration of 287. Sodium atoms have 11 protons and so 11 electrons.
Indicates the two fundamental spin states of an electron in an orbital electron configuration a list of all the electrons in an atom or ion. The chemical inactivity of noble gas atoms reflects the stable arrangement of their electrons one which other atoms cannot easily disturb. The arrangement of electrons in an atom effects the chemical properties of the elements.
The energy of atomic orbitals increases as the principal quantum number n increases. Orbital Energies and Atomic Structure. This electron arrangement can be written as 2.
An electron will move to the orbital with lowest energy. Rules to predict electron configuraon. Generally electrons are equal to protons but in ions it is different so use protons.
Electrons occupy the lowest energy orbitals available first. Sodium atoms have 11 protons and so 11 electrons. Answer 1 of 2.
We will start with a very simple way of showing the arrangement of electrons around an atom. Has an atomic number of 2 8 1 11. The chemical behavior of an element can be understood by looking at the electron assignment of an atom of the element as compared to the electron assignments of neighboring noble gas atoms.
The examples below show the electron arrangement of some elements. The next atom is helium with 2 electrons. The formula helps in determination of arrangement of electrons and is.
What does the arrangement of electrons in an atom determine. What is electron arrangement in chemistry. To build up electron configurations for the ground state of any particular atom first the energy levels are determined then electrons are added to the orbitals one by one according to three.

Great Blog For Hs Science Chemistry For Kids Homeschool Science Chemistry

Is Nh2 Polar Or Non Polar Amide Ion In 2021 Molecules Nh 2 Electrons

Electron Configuration For Te Tellurium In 2021 Electron Configuration Electrons How To Find Out

Bromine Electron Configuration Electron Configuration Element Chemistry Element Symbols

Atomic Structure Worksheet Atomic Structure Chemistry Worksheets Worksheets

Atomic Structure Worksheet Chemistry Classroom Chemistry Worksheets Teaching Chemistry

Is Pcl5 Polar Or Nonpolar Phosphorous Pentachloride In 2021 Molecular Geometry Molecular Molecules

Electron Configuration Boundless Chemistry

12 Mg Magnesium Electron Shell Structure Schoolmykids Electron Configuration Element Chemistry Magnesium

Electron Arrangement In Atom Spm Chemistry
Electron Configuration With Examples Online Chemistry Tutorials

Periodic Table Of Elements Activity Atomic Structure And The Periodic Table Game Middle School Science Classroom Chemistry Activities Middle School Science

Electron Arrangement In An Atom Youtube
Posting Komentar untuk "Arrangement Of Electrons In An Atom Determine"