Arrangements Of Electrons In An Atom
As shown in the figure below there are multiple energy levels where electrons can be found. If there are more electrons than protons the ion has a negative charge and is called an anion.

Electron Arrangement In An Atom Electrons Teaching 4th Grade Science
The 4s sublevel is lower in energy than the 3d sublevel.

Arrangements of electrons in an atom. Fixed energy value an electron in an atom may have. However Bohr model failed to predict the observed spectra for atoms with more than one. Working out the number of electrons in an atom.
The fourth can also go in 2s but remember about the opposing spins 1s 2 2s 2. Therefore the electron configuration for helium atoms is 1s 2 spoken as one-ess-two. The 1s subshell cannot hold 3 electrons because an s subshell can hold a maximum of 2 electrons.
Some of these microstates have the same energy are degenerate whereas others have different energy. Third orbit has the balance of 4 electrons partially filled. This electron arrangement can be written as 2.
To designate the electron configuration we use the level number and the letter of the sublevel and a superscript number to represent the number of electrons contained in the sublevel. Electron Arrangement in Atoms 1 Electron Arrangement In an atom electrons and the nucleus interact to make the most stable arrangement possible. The atoms of all group 1 elements have similar chemical properties and reactions because they all have one electron in their outer.
By understanding the energy levels of electrons in an atom we can predict properties and understand behavior of the atom. Therefore the electron configuration for helium atoms is 1s 2 spoken as one-ess-two. Elements are shown from atomic number 1 hydrogen up to 94.
It appears discontinuous as the electron jumps from one energy level to another in a few nanoseconds or less. Electrons fill the lowest available sublevels first according to the Aufbau principle. The main idea behind electron arrangements is that electrons can only exist at certain energy levels.
Arrangements of electrons in polyatomic atoms for an atom with several valence electrons a number of arrangements of these electrons in orbitals of different l and ml are possible. Bohrs Explanation for the Emission Line Spectrum of Hydrogen 1 Electrons revolve around the nucleus in fixed paths called Orbits or Energy levels 2 Electrons in any one orbit have a fixed or quantised amount of energy 3 Electrons in an energy level do not gain or lose energy 4 When atoms absorb energy electrons jump from a lower energy level to a higher energy level 5 At these higher levels the electrons. Electron configurations are shorthand descriptions of the arrangements of electrons in atoms.
And we use the Pauli exclusion principle and other rules to arrange the electrons. Sodium atoms have 11 protons and so 11 electrons. Answer 1 of 2.
Electron ShellsIn atomic physics and quantum chemistry electron configuration is the arrangement of electrons of an atom a molecule or other physical stru. Boron Z 5 It has five electrons. Viewed simply electrons are arranged in shells around an atoms nucleus.
The arrangement of electrons in an atom effects the chemical properties of the elements. Electron configurations are shorthand descriptions of the arrangements of electrons in atoms. Although two can go into the 1s orbital the third one must be placed in the 2s.
B Bohrs model of the atom appeared successful in that seemed consistent with observed chemical and physical properties of atoms. Bohr was able to calculate the energy levels of an electron in a hydrogen atom. The electron configuration of a hydrogen atom is spoken out loud as one-ess-one Helium atoms have 2 electrons.
Sodium has a electronic configuration of 281 and Chlorine has an electronic configuration of 287. The arrangement of electrons in atoms. The electron arrangements of the first three elements in group 1.
Take Sodium and Chlorine for example. The electron configuration of a hydrogen atom is spoken out loud as one-ess-one Helium atoms have 2 electrons. Inside an atom the electron occupy only certain positions with a very definite radius from the nucleus energy levels orbits shells.
The period an element is located in is. Therefore the electron configuration for helium atoms is 1s 2 spoken as one-ess-two. The ways electrons are arranged in various orbitals are called electron configuration.
Beryllium Z 4 It has four electrons. Unique to the electron arrangements. Lithium Z 3 It has three electrons.
Arrangements of electrons in the orbitals of an atom is called its electron configuration. 1s 2 2s 1. How are electrons arranged in the atom.
Electron configurations are shorthand descriptions of the arrangements of electrons in atoms. First orbit the inner most energy level has 2 electrons completely filled. Periods are numbered 1-7.
Now as you can see in the third shell of the. In a more realistic model electrons move in atomic orbitals or subshells. ÐThese arrangements are called microstates.
Electrons further away from the nucleus will have higher energy. Both electrons fit into the 1s subshell because s subshells can hold up to 2 electrons. No two electrons in the same atom can have the same set of four quantum numbers Hunds Rule orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron and all electrons in singly occupied orbitals must have the same spin.
The horizontal rows in the periodic table are called periods. Both electrons fit into the 1s subshell because s subshells can hold up to 2 electrons. For example the atomic number of sodium is 11.
The arrangement of electrons in atoms is responsible for the periodicity periodic pattern of the element properties. The number of a period gives information about eh electronic configuration of an element. Both electrons fit into the 1s subshell because s subshells can hold up to 2 electrons.
The atom returns to the ground state as the electrons give off the same amount of energy that they absorbed in the form of light photons electron transition Electrons can change energy levels only with _____ amounts of energy. An ion of an atom is one in which the number of protons and electrons is not the same. An electron occupying these energy levels can only possess very specific amounts of energy.
For example the atomic number of sodium is 11. The 1s subshell cannot hold 3 electrons because an s subshell can hold a maximum of 2 electrons. Electrons closest to the nucleus will have the lowest energy.
If there are more protons than electrons an atomic ion has a positive charge and is called a cation. The electron configuration of a hydrogen atom is spoken out loud as one-ess-one Helium atoms have 2 electrons. Each Silicon atom has its electrons arranged in groups of energy levels or shells as the following.
Atomic electron transition is a change of an electron from one quantum state to another within an atom or artificial atom. Sodium atoms have 11 protons and so 11 electrons. Second orbit has 8 electrons completely filled.
The electron arrangement of an atom can be worked out from its atomic number. This page introduces the way that electrons are arranged in atoms. It looks in detail at the arrangement of the electrons in the first 20 elements in the Periodic Table in energy levels and picks out useful patterns for some of the bigger atoms.
Explain in terms of energy sublevels why the arrangement of electrons in the main energy levels in a calcium atom is 2 8 8 2 and not 2 8 10. The electron arrangement of an atom can be worked out from its atomic number. It is also known as atomic transition quantum jump or quantum leap.

See The Electron Configuration Diagrams For Atoms Of The Elements Electron Configuration Electrons Electron Affinity

The Electron Configuration Of Potassium And Its Atomic Number Atom Model Bohr Model Atom Model Project

Potassium Electron Configuration Potassium Atom Electron Configuration Element Chemistry

Electronic Configurations Intro Chemistry Libretexts Electron Configuration Chemistry Notes Configuration

Electron Configuration Easy Science Electron Configuration Electrons Chemistry Class

See The Electron Configuration Diagrams For Atoms Of The Elements Electron Configuration Atom Diagram Electrons

Learning Card For Electronic Configuration In An Atom Electron Configuration Middle School Science Teacher Atomic Structure

See The Electron Configuration Diagrams For Atoms Of The Elements Potassium Atom Electron Configuration Atom Diagram

Electron Shell Electron Configuration Scientific Notation Word Problems Chemistry Worksheets
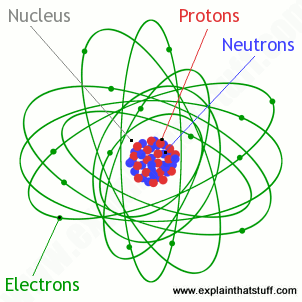
Atoms What Are They What S Inside Them Explain That Stuff Proton Neutron Electron What Is Atom Atom

12 Mg Magnesium Electron Shell Structure Schoolmykids Electron Configuration Element Chemistry Magnesium

Bromine Electron Configuration Electron Configuration Element Chemistry Element Symbols

6 C Carbon Electron Shell Structure Schoolmykids Carbon Element Electron Configuration Element Chemistry

See The Electron Configuration Of Atoms Of The Elements Neon Atom Electron Shell Diagram Atom Diagram Electron Configuration Neon Atom
Posting Komentar untuk "Arrangements Of Electrons In An Atom"