Describe The Motion And Arrangement Of Molecules In A Liquid
C22A Describe conduction in terms of molecules bumping into each other to transfer energy. They are less tightly packed than in solids that is they have bigger intermolecular spaces.

Online Activity Particles In Solids Liquids And Gases Students View Animation Of Water And Air Partic Matter Science Online Activities School Inspiration
In any fluid the molecules themselves are in constant random motion colliding with each other and with the walls of any container.

Describe the motion and arrangement of molecules in a liquid. Quick Really Spread Out And FastExplanation. C43B Recognize that solids have a more ordered regular arrangement of their particles than liquids and that liquids are more ordered than gases. 12-3 Objectives C22B Describe the various states of matter in terms of the motion and arrangement of the molecules atoms making up the substance.
____definite shape and volume particles are close together they are fixed in place but can vibrate. This force of attraction known as the intermolecular force of attraction holds the molecules together. Solid vibrate jiggle but generally do not move from place to place.
Gas vibrate and move freely at high speeds. -Has a definite volume but no definite shape. Particles are closely connected in a crystal lattice vibrate slightly on the spot.
In liquid since the particles are little far away from each other and possess more kinetic energy than solid thus they can move freely and glide across each other. MS-PS1-4 In a liquid the molecules are constantly in contact with others. Molecules in a gas have the greatest degree of motion.
-Has a definite shape and volume. The heat provided to the water weakens and breaks the bonds between the particles causing it to spread out much easily- Evaporate making steam. _____definite volume but no definite shape particles are close together but can slide freely past on another.
Liquids and solids are often referred to as condensed phasesbecause the particles are very close together. The motion of fluids and the reaction to external forces are described by the Navier-Stokes Equations which express a conservation of mass. Particles roll over one another can move around some space between particles.
Constantly moving spread out a bit Gas. Like solids liquids are capable of vibrational motion but at the same time they can also show rotational and translational motions due to weak intermolecular forces between molecules. The molecules of liquids.
The water molecules in the steam produced when liquid water is evaporated would be very loose and spread out very quickly due to the very weak molecular forces. Describe the motion and arrangement of molecules in a liquid. 146 understand how to use dot-and-cross diagrams to represent covalent bonds in.
In a gas they are widely spaced except when they happen to collide. In a solid atoms are closely spaced and may vibrate in position but do not change relative locations. The bonds are constantly breaking and reforming so the liquid can flow.
Label which phase or phases of the substance is present in each of the three portions of the heating curve. This also keeps the liquid at a constant volume. Liquid vibrate move about and slide past each other.
Liquids have a definite volume but they dont have a definite shape. In solids the molecules are tightly packed with each other with strong force of attraction thus they are not free to move but vibrates at their own position. Describe the arrangement and motion of the molecules during each portion of the graph.
Solids have a definite shape and volume. The molecules in gases are spread apart and move very freely with very much energy. Molecules of a liquid have more freedom of movement than those in a solid.
Because liquid molecules can move their shape is not fixed they tend to. The molecules in a liquid can move about a little. Next Post Next A mass of 030kg is attached to a spring and is set into motion with a period of 024s.
Diatomic molecules including hydrogen oxygen nitrogen halogens and hydrogen halides inorganic molecules including water ammonia and carbon dioxide organic molecules containing up to two carbon atoms including methane ethane ethene and those containing halogen atoms. In a liquid the molecules have more energy and they move around more freely. Describe the arrangement and movement of the octane molecules when it is a liquid Describe the arrangement and movement of the octane molecules when it is a liquid.
Gases do not have a definite shape or volume. Due to the movement of molecules in a liquid the molecules in a liquid are constantly in motion even if the liquid as a whole is stationary the arrangement of molecules in the liquid is irregular. Particles have more energy than those in a solid but less than those in a gas.
Molecules inside liquids are loosely bonded to their neighboring molecules. Arrangement of Molecules in Solid Liquid and Gas - YouTube. Describe the motion and arrangement of molecules in a solid.
Gases and liquids are made of molecules or inert atoms that are moving about relative to each other. Particles have the most energy shown by the diagram as the particles are the most spread apart motion is more random and frequent. All molecules are in constant motion.
Sketch the heating curve for a larger sample of the same liquid. Hence liquids can show random molecular motions but less random compared to gas molecules. Describe the structure of solids liquids and gases in terms of particle separation arrangement and types of motion.

Arrangement Of Molecules In The Three States Of Matter States Of Matter Interactive Science Notebook Middle School Science Experiments

How Sound Travels Solid Liquid Gas Interactive Teaching Sound Teaching Chemistry Science
How Are Molecules Arranged In The Three States Of Matter Quora
How Are Molecules In A Solid Different From Molecules In A Liquid Quora

Difference Between Solid Liquid And Gas In Tabular Form States Of Matter Simplified Solid Liquid Gas Intermolecular Force States Of Matter

Grades 11 12 Nsdl An Enormous Variety Of Biological Chemical And Physical Phenomena Can B States Of Matter Matter Science Middle School Science Experiments

2 1 Simple Kinetic Molecular Model Of Matter Flashcards Quizlet

8 Kinetic Model Of Matter Content States Of Matter Brownian Motion Kinetic Model Solid Liquid Gas Science Blog Physics Tuition
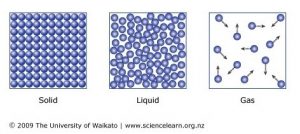
5 10 Describe The Arrangement And Motion Of Particles In Solids Liquids And Gases Tutormyself Chemistry

Pin By Katie Mccuddin On Grammar Matter Experiments States Of Matter Whole Brain Teaching

Brownian Motion Wikipedia The Free Encyclopedia Brownian Motion Motion Philosophy Of Science

Molecules Matter Chapter 1 Matter Solids Liquids And Gases Middle School Chemistry Middle School Chemistry Molecules Middle School Lesson Plans

Pin By Megan Escobar Olsen On Teaching Middle School Science Matter Science Elementary Science Science Lessons

This Lesson Is Designed For The New 2019 2021 0653 Igcse Combined Science Course The Presentation States Of Matter Matter Science States Of Matter Worksheet
Posting Komentar untuk "Describe The Motion And Arrangement Of Molecules In A Liquid"