Arrangement And Movement Of Particles In Solid Liquid And Gas Brainly
Takes the shape of the container. Gas are well separated with no regular arrangement.

Arrangement Of Particles In Phases Of Matter Comparison Expii
Gas are well separated with no regular arrangement.

Arrangement and movement of particles in solid liquid and gas brainly. Far away from each other in gas. The three states of matter. The arrangement of particles in solids liquids and gases - Edukite Learning - YouTube.
Liquids are difficult to compress as particles have less space between them to move. Gases liquids and solids are all made up of microscopic particles but the behaviors of these particles differ in the three phases. Answer 1 of 4.
More than solids but less than. Example of a liquid state of matter. Liquid vibrate move about and slide past each other.
Particle theory of solids liquids and gases explained including the movement and arrangement of particles and how this relates to the properties of solids. Liquid are close together with no regular arrangement. Solid Liquid or Gas.
Particles in a solid are still and have no energy the particles arent moving. Solid Liquid or Gas. The particle model represents particles by small solid spheres.
Particle Arrangement Solid Definite volume and shape Vibrates in place Very low Liquid Definite volume no definite shape Slips and slides past each other moves more quickly than a solid low Gas No definite volume or shape Moves freely to fill its container High Plasma No definite volume or shape Very fast many collisions between particles Very high. Very little Liquid State. As the particles dont move from their position all solids keep a definite shape and its volume stays the same and cannot flow like liquids.
The arrangement of particles in solids liquids and gases - Edukite Learning. No arrangement at all. The particles in a liquid are close together touching but they are able to.
They come together and form droplets of liquid water. Are very strongly attracted to each other. Particles are mainly touching but with some gaps Less dense than solids.
Very slow movement of particles. The reverse can also occur. In solid they closed to each no space between them.
Particles are much further apart Almost no forces of attraction between particles. The rate of diffusion in liquids is higher than that of solids. Changes Between Solids and Gases.
Are solid liquid and gas. Solid Liquid or Gas. Solid Liquid or Gas.
Force of attraction between the particles is weaker than solids. What about a foam. Particles in a gas move freely and have lots of energy a high energy level.
Force of attraction between the particles is negligible and they can move freely. Liquid slow than gases. Solid are tightly packed usually in a regular pattern.
Particles in a liquid are moving and can flow to fill and fit a space the particles have some energyLike solids all of the particles are touching. This process in which a gas changes to liquid is known as condensation. Solids cannot easily be compressed because the particles are already packed very closely together.
In gases particles are far apart from each other. Are closely packed in a fixed arrangement. All of the particles in a solid are touching and are compact.
The molecules are in constant contact with its neighbor generally in a regu. In respect to this what is the movement of a solid. Although we cannot see it or feel it the particles are moving vibrating in place.
Have medium kinetic energy. Close and touching Arrangement. Sound is slower in liquids and slowest in gases.
Solid Liquid or Gas. However sometimes solids change directly to gases and skip the liquid state. The model is used to explain the physical properties of solids liquids and gases.
Gas vibrate and move freely at high speeds. Watch this animation to learn about the arrangements of molecules in a substance and how it decides the states of matter. Water Spacing between the particles.
Liquid lower closer to each other. Solids that change to gas passes through the liquid state first. Solid are tightly packed usually in.
Movement of particles. Random close arrangement of particles. The bonds in a liquid are strong enough to keep the particles close together but weak enough to let them move around each other.
Sound travels the fastest in solids like ironsteel etc. Force between particles is less effective as particles can move Gas. Move around each other sliding but always touching Energy.
The molecules are moving at random through space occasionally colliding but general separated by several diameters at atmospheric pressure. Particles are closely and regularly packed The particles vibrate Strong forces of attraction between particles Liquid. Liquid are close together with no regular arrangement.
Vibrate in their fixed positions but do not move apart Energy. It describes the arrangement movement and energy of particles in a substance. Close and touching Arrangement.
Have low kinetic energy. The particles in a solid are tightly packed and locked in place. The particle theory of matter is a model that describes the arrangement and movement of particles in a substance.
In this video you are shown what the particles are doing in a solid liquid and a gas. Water milk blood coffee etc. Liquids have fixed volume but no fixed shape.
Solid Liquid or Gas. The particle model describes the energy arrangement and movement of particles in solids liquids and gases. Ice Spacing between the particles.
The particles in a solid cannot move around but they can only vibrate from side to side. Has a definite volume. That implies that sound travels fast in a denser medium that is more the density of a medium more is the speed of sound in that medium.

Activity 2 Explaining Arrangements And Movement Of Particles In Solid Liquid And Gas Brainly Ph

Arrangement Of Molecules In Solid Liquid And Gas Youtube

Conclusion For Model Demonstrating Movement Of Particles In Solids Liquids And Gases Brainly In
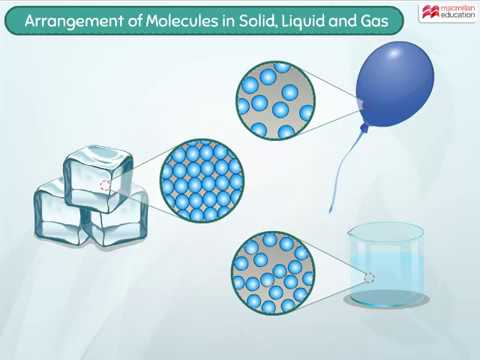
Arrangement Of Molecules In Solid Liquid And Gas Youtube

6 2 Solids Liquids And Gases Particle Model Of Matter Siyavula

Arrangement Of Particles In Phases Of Matter Comparison Expii

Descriptionsolidliquidgasarrangement Of Particlesmovement Of Particlesforces Between Particlesspaces Brainly Ph

Complete The Table How Do Particles Of Solid Liquid And Gas Differ Brainly Ph

Arrangement Of Particles In Phases Of Matter Comparison Expii
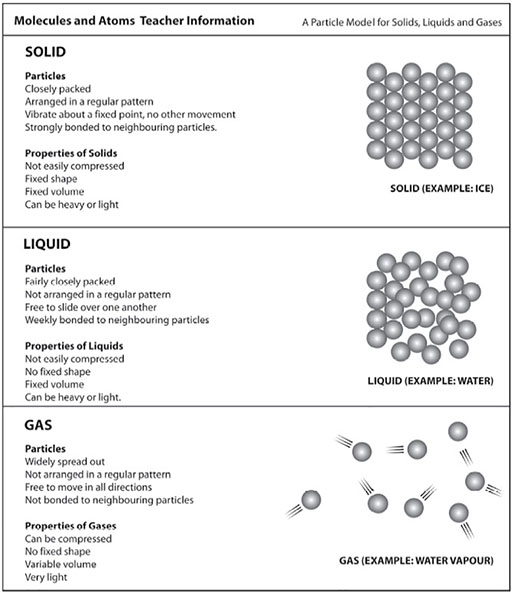
Module 2 Science Investigating Materials View As Single Page

Draw Diagram Showing The Arrangement Of Particles In Solid Liquid And Gases Brainly In

6 1 What Is The Particle Model Of Matter Particle Model Of Matter Siyavula

Activity 3 Illustrating Arrangement And Movement Of Particles In Solid Liquid And Gasobjectiveat Brainly Ph

The Arrangement Of Particles In Solids Liquids And Gases Edukite Learning Youtube
Posting Komentar untuk "Arrangement And Movement Of Particles In Solid Liquid And Gas Brainly"