Arrangement Of Electrons In An Atom Called
Arrangements of electrons in the orbitals of an atom is called its electron configuration. These electrons can rotate around the nucleus in all directions.
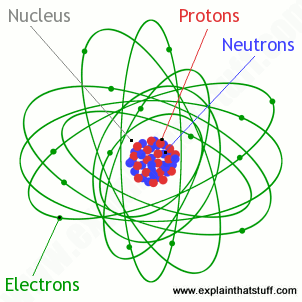
Atoms What Are They What S Inside Them Explain That Stuff Proton Neutron Electron What Is Atom Atom
The number of electrons and protons are same in any atom thus making it neutral or uncharged.

Arrangement of electrons in an atom called. That means that it has 11 protons and. The examples below show the electron arrangement of some elements. The way the electrons are arranged in an atom is called the atoms electronic structure or electronic.
We describe an electron configuration with a symbol that contains three pieces of information. Consider the unbalanced chemical equation below. In atomic physics and quantum chemistry the electron configuration is the distribution of electrons of an atom or molecule or other physical structure in atomic or molecular orbitals.
The way in which the electrons are distributed in the shells of an atom is called the electron arrangement of the atom. The electron configuration of 118. And we use the Pauli exclusion principle and other rules to arrange the electrons.
The arrangement of electrons in an atom is called the atoms ______________________. 1s 2s 2p 3s 3p 4s 3d 4p notice how the 3d comes before 4p. The arrangement of electrons in different orbits and orbitals of an atom in a certain order is called electron configuration.
Lithium has 3 protons and 3 neutrons and three electrons as well. The electrons in an atom move around the nucleus in regions known as electron shells. The examples below show the electron arrangement of some elements.
The number of protons present in an atom specifies its atomic number. And we use the Pauli exclusion principle and other rules to arrange the electrons. Lithium Li has an atomic number of 3 meaning that in a neutral atom the number of electrons will be 3The energy levels are shown as concentric circles around the central nucleus and the electrons are placed from the inside out.
0 1s22s22p 1s22s22p 3s23p O 1522s22p63523p64523d1046 оо 1. The way in which the electrons are distributed in the shells of an atom is called the electron arrangement of the atom. CaSiO3 s HF g - CaF2 aq SiF4 q H20 1 Suppose a 315 g sample of CaSiO3 reacted with 315 L.
Each electron shell can only contain a certain number of electrons. The corresponding numbers of electrons are arranged in different elliptical orbits called shells around the nucleus. The energy of an electron in an atom cannot just have any value but is restricted to having only certain values or levels of energy.
The arrangement of electrons in a lithium atom. The number of protons is the atomic number which you can find from a Periodic Table. The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom.
Working out the number of electrons in an atom. Electrons in an atom tend to assume the arrangement that gives the atom the _______________ possible energy. And they have a.
The electrons present in the outermost shell of an atom are known as the valence electrons. From the Bohr-Bury scheme we find out the outermost shell of an atom can accommodate a maximum of 8 electrons and thus it was observed that the elements having a completely filled outermost shell meaning zero valency show little chemical activity. Chemistry of Structure of an Atom.
Ground state electron configurations can be predicted by a strict set of rules known as the Aufrau principle aufbaumeans filling up. To write the ground state electronic configuration of an atom electrons are added to the lowest energy orbitals giving each orbital two electronsThis is called Aufbau Principle. Tive pole 2 the oxygen end which is the positive pole 3 the hydrogen end which is the negative pole 4.
Electrons are placed in the lower energy level first and when this is completely filled we move on to the second energy level and so on. The order of filling of orbitals in atoms it is different for positive ions is. So if say you are interested in sodium Na you will find that it has an atomic number of 11.
Arrangements of electrons in the orbitals of an atom is called its electron configuration. Rules for Determining Electron Configuration of Atom Rule 1. The notation specific to the organization or arrangement of electrons within orbitals surrounding the nucleus of an atom is called electron configuation.
What is the arrangement of electrons within an atom and it describes how the electrons are distributed among orbitals. The way in which the electrons are distributed in the shells of an atom is called the electron arrangement of the atom. Generally the higher the energy of a shell the farther it is on average from the nucleus.
What is the electron configuration for a neon atom Ne in its ground state lowest energy state. Remember that electrons carry a 1- charge and protons carry a 1 charge. What is the arrangement of electrons in an atom called.
The lowest potential energy arrangement of electrons in an atom is called the ground state. Orbits fixed energy levels. Electrons are arranged in the orbits of the atom at certain distances and certain rules around the atom.
The hydrogen end which is the positive pole Submit Answer. They are also called. An electron arrangement is the way in which electrons are arranged in an atom.
The nucleus center of the atom contains the protons positively charged and the neutrons no charge. For example the electron configuration of the neon atom is 1s2 2s2 2p6. This arrangement of atoms is the most ______________ arrangement.
The outermost regions of the atom are called electron shells and contain the electrons negatively charged. Atoms have different properties based on the arrangement and number of their basic particles. When you draw the arrangement of electrons in an atom out you use orbital box.
Lithium has 3 protons and 3 neutrons and three electrons as well. To designate the electron configuration we use the level number and the letter of the sublevel and a superscript number to represent the number of electrons contained in the sublevel. Lithium has 3 protons and 3 neutrons and three electrons as well.
Electrons are organized according to their energies into sets called shells A grouping of electrons within an atom. The arrangement and distribution of electrons in different orbits was given by Bohr and Bury. The examples below show the electron arrangement of some elements.
When an atom has a full shell of electrons it is considered to have an octet or pseudo-noble gas configuration. Electrons revolve around fixed paths called. Lets look at building up the electronic arrangement electron configuration from hydrogen Z 1 as far as krypton Z 36.
In a neutral atom there must be equal numbers of protons and electrons.

Electron Arrangement In An Atom Youtube

12 Mg Magnesium Electron Shell Structure Schoolmykids Electron Configuration Element Chemistry Magnesium

Rutherford Model Definition Facts Atomic Structure Atom Neon Atom

4 2 Structure Of Atoms Spm Science Atomic Structure University Physics Atom

Electron Configuration Ppt Video Online Download

Electronic Configurations Chemwiki Electron Configuration Text Features Worksheet Complex Sentences Worksheets

Atom Structure By Cnx Openstax Cc By 4 0 Https Creativecommons Org Licenses By 4 0 Atom Activities Electron Configuration Atom

The Electron Configuration Of Potassium And Its Atomic Number Atom Model Bohr Model Atom Model Project

Electron Configuration Boundless Chemistry

Boron Electron Configuration And Full Orbital Diagram In 2021 Electron Configuration Electrons Ionization Energy

Learning Card For Electronic Configuration In An Atom Electron Configuration Middle School Science Teacher Atomic Structure

Potassium Atom Showing Electrons In Their Shells Gcse Chemistry Electrons Potassium Atom

6 C Carbon Electron Shell Structure Schoolmykids Carbon Element Electron Configuration Element Chemistry

Arrangement Of Electrons In An Atom Structure Of Atoms Cbse Grade 9 Chemistry Youtube
Posting Komentar untuk "Arrangement Of Electrons In An Atom Called"