Describe The Arrangement And Movement Of Molecules In Liquid Water
The free electrons may move around inside the substance. In a gas they are widely spaced except when they happen to collide.
How Are Molecules Arranged In The Three States Of Matter Quora
- Water vaporWater molecules move freely with much more space between them than in the liquid of solid state.

Describe the arrangement and movement of molecules in liquid water. In the liquid phase the molecular forces are weaker than in a solid. Ice liquid water and water vapor differ in the arrangement and motion of water molecules. Even in its tetrahedral form liquid water is different from ice because the bonds are constantly breaking the molecules of water moving and more bonds forming.
Because the rigid open structure of ice has broken down water molecules can crowd closer together. In a liquid the molecules have more energy and they move around more freely. Liquids have a definite volume but they dont have a definite shape.
-Has a definite shape and volume. In microgravity a liquid forms a ball inside a free surface. Water molecules are locked in rigid pattern and vibrate in their places.
In a solid the atoms are tightly packed together meaning that they vibrate in place slowly. Describe the arrangement and movement of the octane molecules when it is a liquid. Gases do not have a definite shape or volume.
They are less tightly packed than in solids that is they have bigger intermolecular spaces. The oxygen atoms in distorted water molecules have 2 strong bonds to hydrogen and 2 weak ones. When water goes solid freezes the molecules get locked together to form the solid phase.
Electrons posses kinetic energy which is not sufficient for them to leave the surface liquid. Thus liquid water is denser than ice. Describe the arrangement of molecules in liquid water and in ice.
Are fewer and more disordered than those between molecules of ice at the same temperature. Particles are closely connected in a crystal lattice vibrate slightly on the spot. The molecules cannot move randomly.
146 understand how to use dot-and-cross diagrams to represent covalent bonds in. Particles roll over one another can move around some space between particles. Regardless of gravity a liquid has a fixed volume.
MS-PS1-4 In a liquid the molecules are constantly in contact with others. Can fill any shape. These molecules cant move around like they can in liquid water.
Gases and liquids are made of molecules or inert atoms that are moving about relative to each other. In a liquid the energy gets faster and they have enough space between each other where they can move past one another. Solids have a definite shape and volume.
Particles can move all over the place spread far apart. The molecules in a liquid can move about a little. The molecules of liquids do not have fixed positions.
0 o C 0oC 0 o C. In the gas phase the molecular forces are very weak. Liquid water molecules shows that the hydrogen bonds between molecules of liquid water at.
Diatomic molecules including hydrogen oxygen nitrogen halogens and hydrogen halides inorganic molecules including water ammonia and carbon dioxide organic molecules containing up to two carbon atoms including methane ethane ethene and those containing halogen atoms. The oxygens in tetrahedral ice-like water have 4 equivalent bonds to hydrogen. Water molecules are much farther apart than in liquid water.
In a solid atoms are closely spaced and may vibrate in position but do not change relative locations. And in a gas the particles are moving very fast and they are spread out even further than a liquid. As here in liquid state water molecules are arranged with no regular movements in solids these molecules are tightly packed usually in a regular pattern and in gaseous state these are well separated with no regular arrangements.
The molecules in gases are spread apart and move very freely with very much energy. A liquid will take the shape of its container with a free surface in a gravitational field. - Liquid waterWater molecules slide and bump past each other.
The intermolecular forces of attraction between the molecules of a liquid. -Has a definite volume but no definite shape. So in different states these molecules have different arrangement.
Tightly or closely packed molecules with no or slight intermolecular space. A gas fills its container taking both the shape and the volume of the. Makes less dense than liquid water.

The Arrangement Of Particles In Solids Liquids And Gases Edukite Learning Youtube

Solids Liquids Gases Density Thermal Energy Insulation Flashcards Quizlet

How To Draw Molecular Arrangements Is Solids Liquids And Gases Solid Liquid And Gases Drawing Youtube
How Are Molecules Arranged In The Three States Of Matter Quora

Arrangement Of Particles In Phases Of Matter Comparison Expii

Arrangement Of Molecules In Solid Liquid And Gas Youtube

Arrangement Of Particles In Phases Of Matter Comparison Expii
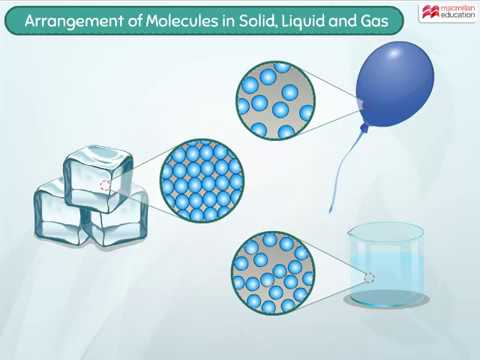
Arrangement Of Molecules In Solid Liquid And Gas Youtube

6 2 Solids Liquids And Gases Particle Model Of Matter Siyavula

It S Just A Phase Modeling The Phases Of Water Ucar Center For Science Education
Posting Komentar untuk "Describe The Arrangement And Movement Of Molecules In Liquid Water"