The Arrangement Of Electrons In An Atom Is Known As The Atoms
Rutherford finds that an atom has a small dense positively charged nucleus. Keeping the Bohr and Bury rules in mind let us write the electronic configuration of elements.
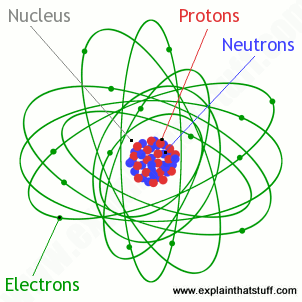
Atoms What Are They What S Inside Them Explain That Stuff Proton Neutron Electron What Is Atom Atom
The way in which the electrons are distributed in the shells of an atom is called the electron arrangement of the atom.

The arrangement of electrons in an atom is known as the atoms. Electronic Configuration of an ElementThe arrangement of electrons in the various shellsorbitsenergy levels of an atom of the element is known as electronic. Remember that electrons carry a 1- charge and protons carry a 1 charge. Max number of electrons in 1st energy level2 n² 2.
It shows that you cannot stand in between two rungs of a ladder you will be magically floating like a electron that can be in one orbit or another but not in between. It is the orbital arrangement of electrons in an atom molecule or other physical structures. The way in which the electrons are distributed in the shells of an atom is called the electron arrangement of the atom.
These scholars imagined atoms as varying in shape depending on the type of atom. Atoms are tiny indestructible particles. In Niels Bohrs model the electron moves in a circular orbit around the nucleus.
For ExFor 1st energy level n1. Niels bohr agreed with rutherfords model of the atom. Each successive shell is further from the nucleus and has a greater energy.
Working out the number of electrons in an atom. Rutherfords Model of the Atom. The arrangement of the electrons in an atom is known as its electron.
The formula helps in determination of arrangement of electrons and is known as Bohr Bury Schemes. Aufbau principle According to the _________ an electron occupies the lowest possible energy orbital that it can receive. Sub shells are known by letters s p d and f.
Formula 2n 2 helps in the determination of the maximum number of electrons present in an orbit here n orbit number. Of electrons that the 3rd shell can contain is 8. The electrons present in the outermost shell of an atom are known as the valence electrons.
Lithium has 3 protons and 3 neutrons and three electrons as well. 1 point giving brainliest for sure There are two electrons in the first energy level and seven electrons in the second energy level. The examples below show the electron arrangement of some elements.
How does it model it. From the Bohr-Bury scheme we find out the outermost shell of an atom can accommodate a maximum of 8 electrons and thus it was observed that the elements having a completely filled outermost shell meaning zero valency show little chemical activity. The maximum number of electrons which can be accommodated in any energy level of an atom is given by 2n²Bohr-Bury Rule.
The distribution of electrons in an atom is called as Electronic Configuration. The nucleus which is in the center of the atom and contains protons and neutrons and the outer region of the atom which holds its electrons in orbit around the nucleus. In the modern periodic table elements are arranged in order of atomic number in periods and groups.
There are two electrons in the first energy level and five electrons. The electron configuration is the arrangement of electrons in an atom. To build up electron configurations for the ground state of any particular atom first the energy levels are determined then electrons are added to the orbitals one by one according to three.
The number and arrangement of electrons in atoms of an element determine the chemical properties of the element. The simplest model of electrons has them orbiting in shells around the nucleus. This section focuses on the arrangement and behavior of electrons in atoms.
2The arrangement of electrons in various energy levels or shells of an atom is known as electronic configuration of the element. The electron configuration of an atom is the arrangement of electrons in the electron cloud around the nucleus of the atom. Now that you know that electrons have quantum numbers how are they arranged in atoms.
No two electrons in an atom can have the same set of four quantum numbersThis dramatically limits the number of electrons that can exist in a shell or a subshell. An atom is composed of two regions. This is an indication of the different orbitals that are occupied by.
The key to understanding electronic arrangement is summarized in the Pauli exclusion principle. The examples below show the electron arrangement of some elements. The arrangment of electrons in an atom is known as the atoms _____.
The maximum number of electrons that the first shell can contain is 2. Which statement best describes the arrangement of electrons in an atom of fluorine F. Read more about Atomic models and Atomic.
The way the electrons are arranged in an atom is called the atoms electronic structure or electronic configuration. Mendeleev made an early periodic table. Water atoms were smooth and slippery explaining why water was a liquid at room temperature and could be poured.
White Light White light when passed through a prism is broken up into an array of colours Such an array is called a spectrum Thus the spectrum is the spread of colours that come out of the prism. Bohrs Study of Spectra Bohr developed his theory about the arrangement of electrons in atoms by studying what were known as the Spectra of Elements 6. The electron orbits or atomic energy levels in Bohrs model can be compared to the rungs of a ladder.
It looks in detail at the arrangement of the electrons in the first 20 elements in the Periodic Table in energy levels and picks out useful patterns for some of the bigger atoms. Ground-state electron configuration is the energy arrangement of the electrons for each element. Electron arrangement is also known as electron configuration.
For the first 20 elements the max. Of electrons that the first shell can contain is 8. They envisaged iron atoms as having hooks which locked them together explaining why iron was a solid at room temperature.
Electronic arrangements model how electrons are arranged. This model can be further refined by the concept of sub shells and orbitals. Rules for Electron Configuration.
The additional electrons go into the fourth shell before the third shell is completely filled. Know what you are doing before you do it. This page introduces the way that electrons are arranged in atoms.
Bohr Model of the Atom.

6 C Carbon Electron Shell Structure Schoolmykids Carbon Element Electron Configuration Element Chemistry

Arrangement Of Electrons In An Atoms Electron Configuration Middle School Science Electrons

Howstuffworks Electrons And Why Magnets Stick Electron Configuration Physical Chemistry Atom

Learning Card For Electronic Configuration In An Atom Electron Configuration Middle School Science Teacher Atomic Structure

See The Electron Configuration Diagrams For Atoms Of The Elements Electron Configuration Electrons Electron Affinity

17 Cl Chlorine Electron Shell Structure Schoolmykids In 2021 Electron Configuration Element Chemistry Periodic Table

See The Electron Configuration Diagrams For Atoms Of The Elements Potassium Atom Electron Configuration Atom Diagram

Potassium Atom Showing Electrons In Their Shells Gcse Chemistry Electrons Potassium Atom

See The Electron Configuration Diagrams For Atoms Of The Elements Electron Configuration Atom Diagram Electrons

Periodic Table Of Elements Activity Atomic Structure And The Periodic Table Game Teaching Chemistry Middle School Science Classroom Science Chemistry

The Electron Configuration Of Potassium And Its Atomic Number Atom Model Bohr Model Atom Model Project

Electron Configuration Easy Science Electron Configuration Electrons Chemistry Class

Electron Arrangement In An Atom Electrons Teaching 4th Grade Science

See The Electron Configuration Of Atoms Of The Elements Neon Atom Electron Shell Diagram Atom Diagram Electron Configuration Neon Atom
Posting Komentar untuk "The Arrangement Of Electrons In An Atom Is Known As The Atoms"