5. Arrangement Of Electrons Around Atomic Nucleus
Electrons further away from the nucleus will have higher energy. The cloud is more dense where the probability of finding the electron is high.

Chemistry Of Calcium Chemwiki Atom Model Bohr Model Atom Model Project
Credit should be given to Niels Bohr for proposing this theory According to Bohrs model of the atom electrons orbit about the nucleus much like the way planets orbit the sun.

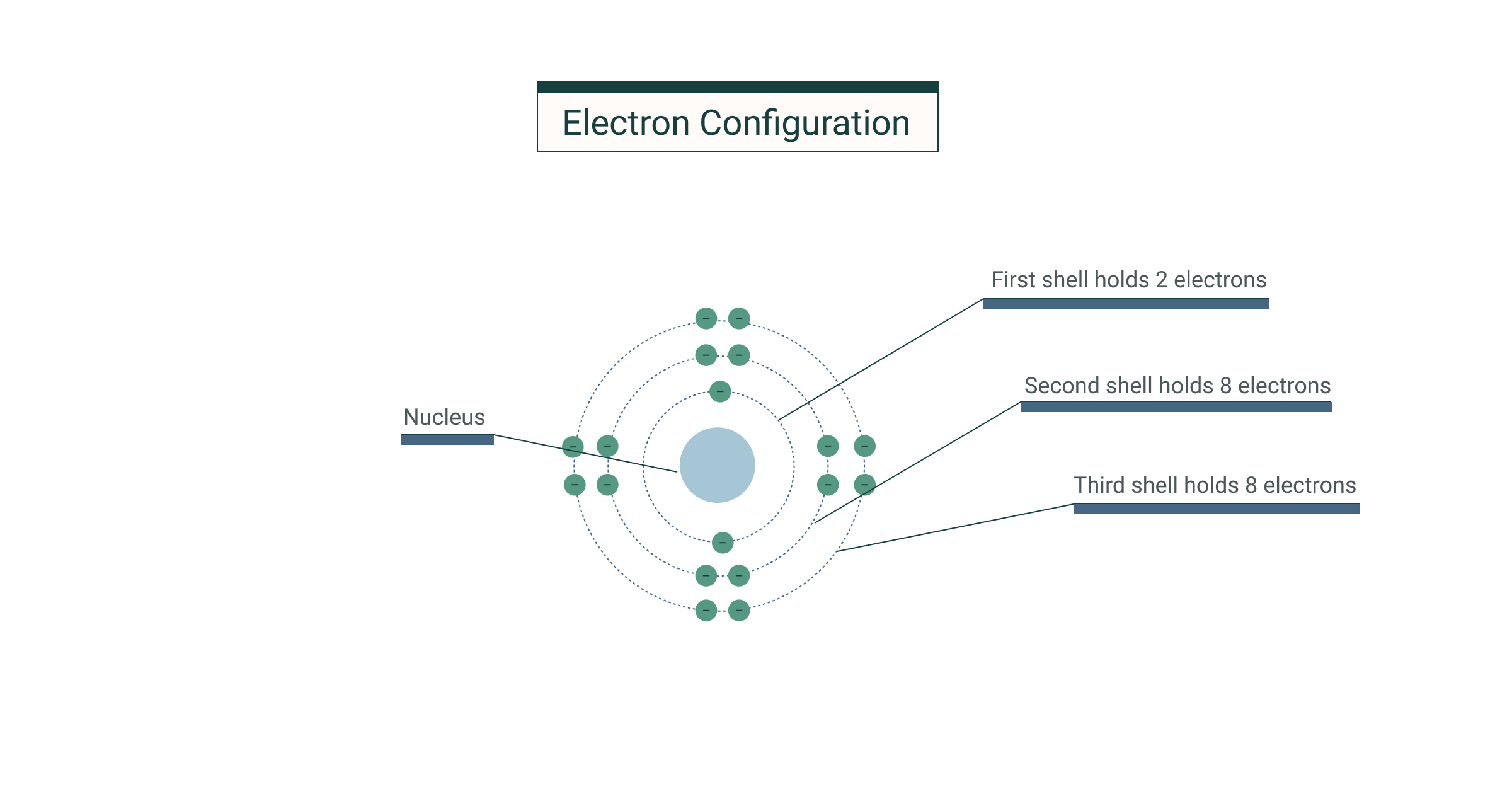
5. arrangement of electrons around atomic nucleus. The shells that are closer to the nucleus are generally lower. Electrons closest to the nucleus will have the lowest energy. Each electron shell can only contain a certain number of electrons.
This chemical behavior is due to the arrangement of e- in atoms. Arrangement of electrons around atomic nucleus. Postulates of Bohrs Atomic Model.
Electrons are negatively charged subatomic particles arranged like a cloud of negative charges outside the nucleus of an atom. An atoms electron shell can accommodate 2n 2 electrons where n is the shell level. Electrons revolve around the nucleus in fixed orbits or energy levels or shells.
In terms of a more refined quantum-mechanical model the KQ shells are subdivided. The electrons in an atom are grouped around the nucleus into shells roughly like the layers in an onion. Primarily the atomic structure of matter is made up of protons electrons and neutrons.
The protons and neutrons make up the nucleus of. Each orbital has at most two electrons. Electrons are always moving around the nucleus and so possess potential and kinetic energy.
The arrangement of electrons around the atomic nucleus is complex and electrons do not simply orbit the nucleus as a planet would orbit a star. An electron with the least amount of energy. Click to see full answer.
ELECTROMAGNETIC RADIATION Form of energy that exhibits wave-like behavior as it travels through space. Arrangement of Electrons in Atoms Honors Chemistry. The arrangement of electrons in the orbitalsrings of an atom.
In the quantum mechanical model it is the probability of finding an electron with a certain volume of space surrounding the nucleus can be represented as a fuzzy cloud. While revolving around the nucleus in an orbit an electron does not lose energy nor does it gain energy. Waves confined around an atomic nucleus.
In atomic physics and quantum chemistry the electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals. The integer n represents the various energy levels 1 2 3. The ground state electron configuration is the arrangement of electrons around the nucleus of an atom with lower energy levels.
Quantum mechanical model of atomic structure. For example the electron configuration of the neon atom is 1s2 2s2 2p6 meaning that the 1s 2s and 2p subshells are occupied by 2 2 and 6 electrons respectively. Electronic configuration also called electronic structure the arrangement of electrons in energy levels around an atomic nucleusAccording to the older shell atomic model electrons occupy several levels from the first shell nearest the nucleus K through the seventh shell Q farthest from the nucleus.
The electrons in an atom move around the nucleus in regions known as electron shells. One electron attracts to both more say 211 proton-electron than the surrounding electron-electron 11 repulsion. That is because electrons for want to be in pairs as far from each other but.
But they can only possess certain values of energy or specific energy levels. Electrons are arrange in shells around the nucleus protons and neutrons and its axis. Viewed simply electrons are arranged in shells around an atoms nucleus.
Atomic theory has affected the field of physics in a way that has given scientist knowledge on how to produce an atomic bomb. The electrons in an atom move around the nucleus in regions known as electron shells. The farther a shell is from the nucleus the larger it is the more electrons it can hold and the higher the energies of those electrons.
In the bohr model the electron is treated as a particle in fixed orbits around the nucleus. There are many types of electromagnetic radiation and all are. In AVSC the electrons lock into places which then gives situations with missing electrons open path to nucleus where the outer electron of another atom can attract both bond.
The electrons occupying the orbitals of varying energy levels naturally falls towards the lowest energy state or ground state. The atomic part of physics is primarily concerned with the arrangement of electrons around the nucleus and the processes by which the arrangements was changed. Answer for the clue The path of an electron around the nucleus of an atom 5 letters.
A region around the nucleus where an electron is likely to be. Tendency of electrons to enter orbitals of lowest energy first. Click to see full answer.
Electrons as waves The electron waves could only exist at specific frequencies that correspond to specific energies- the quantized energies associated with the Bohr orbits. The arrangement depends upon of their potential energies in different orbits. In the qmml the electron is treated mathematically as a wave.
The atomic structure of an element refers to the constitution of its nucleus and the arrangement of the electrons around it. CH 5 ELECTRONS IN ATOMS 5-1 Light and Quantized Energy Some elements emit visible light when heated with a flame. Answer 1 of 5.
Area outside the nucleus the electrons are likely to be found. That electrons be considered waves confined to the space around the atomic nucleus diffraction bending of a wave as it passes by the edge of an object or through a small opening. Electronic configurations describe each electron as moving independently in an orbital in.
Electrons are placed in the lower energy level first and when this is completely filled we move on to the second energy level and so on. Each electron shell can only contain a certain number of electrons. Electrons are placed in the lower energy level first and when this is completely filled we move on to the second energy level and so on.
Broadly speaking electrons are located in concentric shells that surround the nucleus.

Learning Card For Electronic Configuration In An Atom Electron Configuration Middle School Science Teacher Atomic Structure

Electron Configuration Easy Science Electron Configuration Electrons Chemistry Class
Electron Configuration And Structure

Atoms Protons Neutrons And Electrons Anchor Poster Protons Neutrons Atom

How To Write Electron Configurations For Atoms Of Any Element Electron Configuration Electrons Atom

Atoms Its Structure Electronic Configuration Isotopes More Reading Notes Electron Configuration Informational Text Reading Notes

Electron Configuration Boundless Chemistry

Atom Orbits And Energy Levels Britannica

Atomic Structure Parts Of The Atom Bundle Atomic Structure Atom Science Lessons

The Electron Configuration Of Potassium And Its Atomic Number Atom Model Bohr Model Atom Model Project

Existing Atomic Structure Electrons Arrangement Around The Nucleus Download Scientific Diagram

Electronic Configurations Intro Chemistry Libretexts Electron Configuration Chemistry Notes Configuration
Posting Komentar untuk "5. Arrangement Of Electrons Around Atomic Nucleus"