Arrangement Of Electrons Atomic Nucleus
An atom of the alkaline earth metal beryllium with an atomic number of 4 contains four protons in the nucleus and four electrons surrounding the nucleus. After the discovery of the neutron in 1932 models for a nucleus composed of protons and neutrons were quickly developed by Dmitri Ivanenko and Werner Heisenberg.

Electron Configuration Easy Science Electron Configuration Electrons Chemistry Class
The protons and neutrons make up the nucleus of.

Arrangement of electrons atomic nucleus. 31 - Arrangement of Electrons. Also the electrons primarily move within orbitals or sub-shells of an atom. Electrons are organized in such a way such that repulsions between them.
In the modern periodic table elements are arranged in order of atomic number in periods and groups. An atom is composed of a positively. The nuclear shell model is partly analogous to the atomic shell model which describes the arrangement of electrons in an atom in that a filled shell results in greater stability.
However the electrons closest to the nucleus contain lowest energy and the furthest ones have the highest energy. One may also ask what is 2n2 rule. According to the older shell atomic model electrons occupy several levels from the first shell nearest the nucleus K through the seventh shell Q farthest from the nucleus.
Answer 1 of 5. That is because electrons for want to be in pairs as far from each other but still net-2-field attracted to the Bohr radius. An electron arrangement is the way in which electrons are arranged in an atom.
The fourth electron fills the remaining space in the 2s orbital. It looks in detail at the arrangement of the electrons in the first 20 elements in the Periodic Table in energy levels and picks out useful patterns for some of the bigger atoms. The way the electrons are arranged in an atom is called the atoms electronic structure or electronic configuration.
The shells that are closer to the nucleus are generally lower. The atomic nucleus is the small dense region consisting of protons and neutrons at the center of an atom discovered in 1911 by Ernest Rutherford based on the 1909 GeigerMarsden gold foil experiment. In the atomic shell model the central potential around which the electrons orbit is generated by the nucleus.
Likewise people ask whats the electron arrangement. Arrangement of Electrons in Atoms Honors Chemistry. The arrangement of electrons in atoms.
The electrons in an atom move around the nucleus in regions known as electron shells. Thus the electron configuration and orbital diagram of lithium are. Electronic arrangements model how electrons are arranged.
The sum of number of protons and number of electrons present in an atom is called atomic weight. In this way electron waves could exist only at specific frequencies. Nucleons are added to shells which increase with energy that orbit around a central potential.
According to the relationship. Working out the number of electrons in an atom. An atom of boron atomic number 5 contains five electrons.
Quantum mechanical model of atomic structure. This rule of arrangement of electrons according to the shell is known 2n2 rule where n means number of shell. The atomic structure of an element refers to the constitution of its nucleus and the arrangement of the electrons around it.
Confined to the space around an atomic nucleus. What is the arrangement of electrons in an atom called. So think hemispheres for the 4th quantum number.
For example the electron configuration of the neon atom is 1s 2 2s 2 2p 6 meaning that. Electrons as waves The electron waves could only exist at specific frequencies that correspond to specific energies- the quantized energies associated with the Bohr orbits. Primarily the atomic structure of matter is made up of protons electrons and neutrons.
The Pauli exclusion principle states that no two electrons in an atom may have the. The ground-state electronic configuration of Selenium is. Electrons are negatively charged subatomic particles arranged like a cloud of negative charges outside the nucleus of an atom.
We know that the electrons are located outside the nucleus of the atom in mostly empty space. The movement of electrons The electrons are negatively - ve charged particles They revolve around the nucleus with very high speed The electron has a negligible mass relative to that of the proton or the neutron so the mass of the atom is concentrated in the nucleus. The electronic configuration of an element is an arrangement of its electrons in different energy levels around the atomic nucleus.
The most stable arrangement of electrons is. The electrons in an atom are grouped around the nucleus into shells roughly like the layers in an onion. In this section we will learn that associated with this empty space are many different energy levels.
Electronic configuration also called electronic structure the arrangement of electrons in energy levels around an atomic nucleus. The farther a shell is from the nucleus the larger it is the more electrons it can hold and the higher the energies of those electrons. The nucleus of an atom is surrounded by orbitals and on these orbitals electrons are arranged.
This page introduces the way that electrons are arranged in atoms. And the electronic configuration in which the arrangement of electrons is with lower energy levels around the nucleus is termed as ground-state electronic configuration. The arrangement of electrons around the atomic nucleus is complex and electrons do not simply orbit the nucleus as a planet would orbit a star.
Broadly speaking electrons are located in concentric shells that surround the nucleus. Waves confined around an atomic nucleus. The arrangement depends upon of their potential energies in different orbits.
The mode of arrangement of electrons in different shells of an atom. The main for i. In atomic physics and quantum chemistry the electron configuration is the distribution of electrons of an atom or molecule or other physical structure in atomic or molecular orbitals.
Mendeleev made an early periodic table. Electrons are arrange in shells around the nucleus protons and neutrons and its axis.

Electron Shell Electron Configuration Scientific Notation Word Problems Chemistry Worksheets

Great Blog For Hs Science Chemistry For Kids Homeschool Science Chemistry

The Electron Configuration Of Potassium And Its Atomic Number Atom Model Bohr Model Atom Model Project
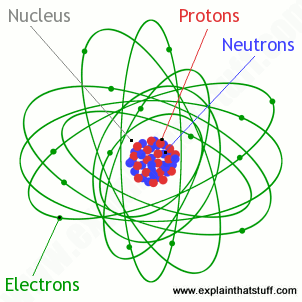
Atoms What Are They What S Inside Them Explain That Stuff Proton Neutron Electron What Is Atom Atom

12 Mg Magnesium Electron Shell Structure Schoolmykids Electron Configuration Element Chemistry Magnesium

4 2 Structure Of Atoms Spm Science Atomic Structure University Physics Atom

Chemistry Of Calcium Chemwiki Atom Model Bohr Model Atom Model Project

Boron Electron Configuration And Full Orbital Diagram In 2021 Electron Configuration Electrons Ionization Energy

Atoms Its Structure Electronic Configuration Isotopes More Reading Notes Electron Configuration Informational Text Reading Notes

Learning Card For Electronic Configuration In An Atom Electron Configuration Middle School Science Teacher Atomic Structure

How Are Electrons Distributed In Different Orbits Electronic Configuration Electron Configuration Electrons Atomic Structure

The Atom Chemistry Is My Jam Planetary Model Electron Configuration Chemistry Classroom

Worldofchemicals On Twitter Atomic Theory Atom Diagram Proton Neutron Electron

Atom Structure By Cnx Openstax Cc By 4 0 Https Creativecommons Org Licenses By 4 0 Atom Activities Electron Configuration Atom
Posting Komentar untuk "Arrangement Of Electrons Atomic Nucleus"