The Arrangement Of Electrons In Energy Levels Around An Atomic Nucleus
The simplest model of electrons has them orbiting in shells around the nucleus. This model can be further refined by the concept of sub shells.

Electron Configuration Boundless Chemistry
They travel very quickly in electron energy levels.

The arrangement of electrons in energy levels around an atomic nucleus. Energy Levels or Shells. Electrons are located outside of the nucleus and arranged by energy levels in the electron cloud. The exact position of an electron in an energy level cannot be said.
An atoms electron shell can accommodate 2n 2 electrons where n is the shell level. This view simply states that electrons are arranged in energy levels or shells around the nucleus of an atom. Sodium atoms have 11 protons and so 11 electrons.
The shells can be denoted by alphabets K L M or quantum numbers. The different energy levels are known as 1 2 3 4. This arrangement follows certain rules known as the Aufbau Hund and Pauli Exclusion Principle.
According to the older shell atomic model electrons occupy several levels from the first shellnearest the nucleus Kthrough the seventh shell Qfarthest from the nucleus. In a more realistic model electrons move in atomic orbitals or subshells. Viewed simply electrons are arranged in shells around an atoms nucleus.
There are a certain number of electrons that each energy level can hold. Arrangement of electrons around atomic nucleus. Inside the shell we have subshells.
The highest energy level n7 is the farthest away from the nucleus. The electrons surrounding an atom are located in regions around the nucleus called energy levels. Electrons further away from the nucleus will have higher energy.
Quantum mechanical model of atomic structure. The electrons revolve around the nucleus in a number of orbits called the energy levels and during their movement they seem to be as a cloud around the nucleus The energy levels are the imaginary regions around the nucleus in which the electrons move according to their energies. The arrangement depends upon of their potential energies in different orbits.
The chemical properties of an element are determined by the arrangement of electrons in its atom. Electrons are arranged in shells or energy levels around the nucleus of an atom. An energy level represents the 3-dimensional space surrounding the.
In chemistry an electron shell or energy level may be imagined as an orbit with electrons around the nucleus of an atom. Each successive shell is further from the nucleus and has a greater energy. The closest shell to the nucleus is called the K shell followed by the L shell then the M shell and so on away from the nucleus.
Electrons are negatively charged subatomic particles arranged like a cloud of negative charges outside the nucleus of an atom. Discrete bundle of electromagnetic energy. Electrons closest to the nucleus will have the lowest energy.
The number of electrons in an atom is equal to the number of protons. The electrons in an atom are grouped around the nucleus into shells roughly like the layers in an onion. The electron shell is known as an energy level present on the outside part of an atom around the atomic nucleus.
In this lesson they will focus on the arrange-ment of the electrons in each element. Mation to understand the basic pattern of the arrangement of electrons on energy levels around an atom. Electrons are found in energy levels around the nucleus of an atom.
These energy levels are numbered 1 2 3 etc. The electron arrangement of an atom can be worked out from its atomic number. Introduce students to the idea that electrons surround the nucleus of an atom in regions called energy levels.
Viewed simply electrons are arranged in shells around an atoms nucleus. The arrangements can be indicated by electron. Electron configuration is the arrangement of electron around the nucleus of an atom based on their energy level.
Electronic configuration also called electronic structure the arrangement of electrons in energy levels around an atomic nucleus. There are a whole lot more energy levels possible in an atom and if you supply enough energy to an atom the electron can jump around between them. Energy levels can also be called shells The energy levels are designated by a principal quantum number n.
Students will be given cards with information about the electrons and energy levels for each of the first 20 atoms. Electrons are arranged in energy levels or shells around the nucleus of an atom. Review with students that in lesson two they focused on the number of protons neutrons and electrons in the atoms in each element.
Each orbital has at most two electrons. Electrons located in the outermost shell of the electron cloud are called valence electrons and have the highest energy. But in the absence of any extra energy the electrons will always be found in the lowest possible energy level - as close as possible to the nucleus.
Sub Shells and Orbitals. These Sub-Shells of an atom are subdivisions of electron shells energy levels represented by s p d f. And the corresponding shells are known as K L M N and so on.
Electrons further away from the nucleus will have higher energy. The lowest energy level is designated as n1 whereas the highest energy level is n7. They will again try to correctly match the cards with each element.
Electrons closest to the nucleus will have the lowest energy. For example the atomic number of sodium is 11. Electrons that are in the first energy level energy level 1 are closest to the nucleus and will have the lowest energy.
From far distances away relative to their size electrons move around the nucleus. The innermost shell lowest energy level which can contain a maximum of two electrons is filled first. Tendency of electrons to enter orbitals of lowest energy first.
The n1 energy level is closest to the nucleus. The farther a shell is from the nucleus the larger it is the more electrons it can hold and the higher the energies of those electrons. Very much like how the planets orbit the sun.
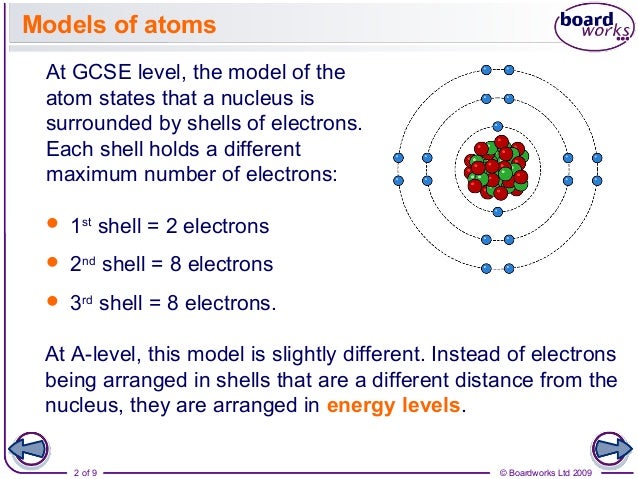
Electron Arrangement Part 1 Energy Levels

Principal Energy Level Easy Science Energy Level Easy Science Science Student

Electron Shell Electron Configuration Scientific Notation Word Problems Chemistry Worksheets

Electron Dot Diagram Easy Science Electron Configuration Electrons Science Facts

Learning Card For Electronic Configuration In An Atom Electron Configuration Middle School Science Teacher Atomic Structure

Electron Configuration Easy Science Electron Configuration Electrons Chemistry Class

What Is Energy Level In Electron Configuration Know It Info

The Electron Configuration Of Potassium And Its Atomic Number Atom Model Bohr Model Atom Model Project

Electron Arrangement Ppt Download

Atom Orbits And Energy Levels Britannica

Electronic Configurations Intro Chemistry Libretexts Electron Configuration Chemistry Notes Configuration
Posting Komentar untuk "The Arrangement Of Electrons In Energy Levels Around An Atomic Nucleus"