Arrangement Of Electrons In An Atom
An electron absorbs energy to jump to a higher energy level. Arrangement of Electrons in Atoms.

12 Mg Magnesium Electron Shell Structure Schoolmykids Electron Configuration Element Chemistry Magnesium
When an electron falls to a lower energy level energy is emitted.

Arrangement of electrons in an atom. Lets look at building up the electronic arrangement electron configuration from hydrogen Z 1 as far as krypton Z 36. Arrangement Of Electrons In Atoms. Pauli Exclusion Principle no two electrons in the same atom can have the same set of four quantum numbers.
The Rutherford model of the atom did not explain why the electron wasnt drawn into the of the atom. The electron arrangement of an atom can be found out from its atomic number. Atoms consist of three basic particles.
A I c 3 b 2 d 4. Development of a New Atomic Model. For example the atomic number of sodium is 11.
Arrangement ofElectrons in Atoms. Read Free Chapter 4 Arrangement Of Electrons In Atoms Answer KeyArrangement of Electrons The lowest energy state of an atom is its ground state. The atomic number of Sulphur is 16 which means that it has 16 protons and 16 electrons where-2 electrons occupy the first shell.
The next atom is helium with 2 electrons. Each electron shell can hold a certain amount of energy. This electron arrangement can be written as 286.
Inside the atom electrons are arranged around the nucleus. The configuration will be as 1s2 2s2 2p5. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.
Electrons in an atom are grouped around the nucleus into shells. The electron shells are shown moving outward from the nucleus. The first orbita l has capacity of two electrons and then the remaining two electrons will be in higher second energy level.
The nucleus center of the atom contains the protons positively charged and the neutrons no charge. This is where the electrons live. This page introduces the way that electrons are arranged in atoms.
All three ways are useful. The electron arrangement of an atom can be worked out from its atomic number. In atomic physics and quantum chemistry the electron configuration is the distribution of electrons of an atom or molecule or other physical structure in atomic or molecular orbitals.
Sodium atoms have 11 protons and. The outermost regions of the atom are called electron shells and contain the electrons negatively charged. Electron Arrangement in An Atom Electron Configuration Atomic Orbital.
1s 2s 2p 3s 3p 4s 3d 4p notice how the 3d comes before 4p. SHORT ANSWER Answer the following questions in the space provided. Displayed as 2n2 n is the energy of the electron.
Orbitals of equal energy are each occupied by one electron before being paired all electrons in singly occupied orbitals have the same spin. _d--_ How many quantum numbers are used to describe the properties of electrons in atomic orbitals. This electron arrangement can be written as 2.
The electron arrangement of an atom can be worked out from its atomic number. 6 electron occupies the third shell. Electron arrangement in the orbitals around an atoms nucleus Aufbau Principle electrons fill orbitals starting at the lowest available possible energy states before filling higher states.
So the second electron could go into the 1s orbital with the opposite spin of the first electron or it could go into the next orbital. This amount is 2 times the amount of energy an electron has squared. Atoms have different properties based on the arrangement.
Working out the number of electrons in an atom. The atoms of all group 1 elements have similar chemical properties and reactions because they all have one electron in their outer. The arrangement of electrons in atoms.
For example the atomic number of sodium is 11. The element atomic number and name are listed in the upper left. Electron configurations are shorthand descriptions of the arrangements of electrons in atoms.
For each electron shell atom diagram the element symbol is listed in the nucleus. For example the electron configuration of the neon atom is 1s2 2s2 2p6. After filling the first orbit the second orbit will consist of 8 electrons.
The electron arrangements of the first three elements in group 1. The atoms of any given element always have the same number of protons. The farther a shell is from the nucleus the larger it is the more electrons it can hold and the higher the energies of those electrons.
The further 5 electrons will be in lower second energy level which is denoted by p. _a--_ A spherical electron cloud surrounding an atomic nucleus would best represent. Protons electrons and neutrons.
ARRANGEMENT OF ELECTRONS IN ATOMS Include graphic organizers for this chapter The following pages contain the bulk but not all of the information for the chapter 4 test Holt mcdougal chemistry. What does the number and arrangement of electrons in. The orbital diagram the electron configuration and the energy diagram.
Early in the 20th century a new model evolved as a result understanding the relationship between light and an atoms electrons. 8 electrons occupy the second shell. So we have three ways to represent the electron arrangement in an atom.
Therefore the electron configuration for helium atoms is 1s 2 spoken as one-ess-two. Bohrs Explanation for the Emission Line Spectrum of Hydrogen 1 Electrons revolve around the nucleus in fixed paths called Orbits or Energy levels 2 Electrons in any one orbit have a fixed or quantised amount of energy 3 Electrons in an energy level do not gain or lose energy 4 When atoms absorb energy electrons jump from a lower energy level to a higher energy level 5 At these higher. An atom always tends to remain in its stable state.
The nucleus is surrounded by electron levels or shells. The electron configuration of a hydrogen atom is spoken out loud as one-ess-one Helium atoms have 2 electrons. Although it can fill up to 18 electrons the 3 rd orbit will fill up to 8 electrons.
The order of filling of orbitals in atoms it is different for positive ions is. The reason behind the arrangement of electrons in such a manner is due to the presence of subshells. Chapter 4 review arrangement of electrons in atoms answer key.
In order for an electron to be ejected from a metal surface the electron must be struck by a single photon with at least the minimum energy needed to knock the electron loose. Electrons in atoms have quantized energies as with the electron on a wire model. It looks in detail at the arrangement of the electrons in the first 20 elements in the Periodic Table in energy levels and picks out useful patterns for some of the bigger atoms.
A grouping of electrons in an atom according to energy. Sodium atoms have 11 protons and so 11 electrons. Both electrons fit into the 1s subshell because s subshells can hold up to 2 electrons.

See The Electron Configuration Diagrams For Atoms Of The Elements Electron Configuration Atom Diagram Electrons

See The Electron Configuration Diagrams For Atoms Of The Elements Electron Configuration Electrons Electron Affinity

Potassium Atom Showing Electrons In Their Shells Gcse Chemistry Electrons Potassium Atom

Electron Shell Electron Configuration Scientific Notation Word Problems Chemistry Worksheets
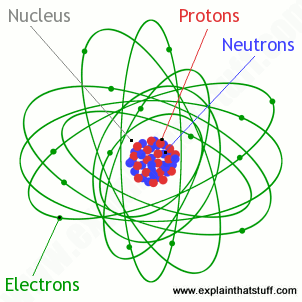
Atoms What Are They What S Inside Them Explain That Stuff Proton Neutron Electron What Is Atom Atom

The Electron Configuration Of Potassium And Its Atomic Number Atom Model Bohr Model Atom Model Project

Bromine Electron Configuration Electron Configuration Element Chemistry Element Symbols

Pin By Elvis Farii On Science Electron Configuration Physical Chemistry Atomic Theory

See The Electron Configuration Diagrams For Atoms Of The Elements Potassium Atom Electron Configuration Atom Diagram

Electron Arrangement In An Atom Electrons Teaching 4th Grade Science

See The Electron Configuration Of Atoms Of The Elements Neon Atom Electron Shell Diagram Atom Diagram Electron Configuration Neon Atom

Potassium Electron Configuration Potassium Atom Electron Configuration Element Chemistry

Learning Card For Electronic Configuration In An Atom Electron Configuration Middle School Science Teacher Atomic Structure

Posting Komentar untuk "Arrangement Of Electrons In An Atom"