K Electron Configuration Long Form
Beryllium Be He 2s 2 2p 1. 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 6 6s 2 4f 14 5d 10 6p 6 7s 2 5f 14 6d 10 7p 6.

Orbital Diagram For Vanadium V Vanadium Electron Configuration Periodic Table
Electron configurations of elements beyond hassium.

K electron configuration long form. The abbreviated form - Ar4s 1 - means the electron configuration of Argon Ar plus one electron in the 4s orbital. Choice 1 is the e-config. Fe 3 1s 2 2s 2 2p 6 3s 2 3p 6 3d 5.
1s2 2s2 2p6 3s2 3p6 4s2 3. The K shell contains a 1s subshell hence it. Electron configuration of Boron B He 2s 2 2p 1.
The electron configuration of bromine is 1s2 2s2p6 3s2p6d10 4s2p5 which can be shortened to Ar 4s2 3d10 4p5. First you should write their normal electron configuration and then when you remove electrons you have to take them from the outermost shell. 1s2 2s2 2p6 3s2 3p6 4.
Fluorine F He 2s 2 2p 6. Electrons have a specific form of distribution or configuration in every atom even Krypton. An online electron configuration calculator provides a condensed method of finding the electron configuration atomic number and atomic mass of given.
It was the first noble gas found to form true. Oxygen O He 2s 2 2p 5. Electron configurations of the elements data page This page shows the electron configurations of the neutral gaseous atoms in their ground states.
When we write the configuration well put all 19 electrons in orbitals around the. Lithium Li He 2s 2. The electronic configuration can quickly and easily tell the reader how many electron orbitals an atom has.
In the case of Rubidium the abbreviated electron configuration is Kr 5s1. None of the above. Nitrogen N He 2s 2 2p 4.
Lithium is the third element with a total of 3 electrons. Nevertheless check the complete configuration and other interesting facts about Rubidium that most people dont know. What is the electron configuration of the cation K.
Electron configuration of Beryllium Be He 2s 2. Atomic number 54 relative atomic mass 131293. Fe 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 6.
Rubidium Overview Rubidium Complete Electron Configuration. Iron has 26 electrons so its normal electron configuration would be. Carbon C He 2s 2 2p 3.
Helium He He 2s 1. Atomic number of K 19 Electronic configuration in short. The full electron configuration of Potassium K is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 1.
Electron configuration of Hydrogen H 1s 1. Concise Form of Electron Configuration Notation Element. All of these shells are filled except the 3p shell which has four electrons.
Therefore th electron configuration for uranium must end as 5f4 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5 p6 6s2 4e14 5d10 6p6 7s2 5f4 This notation can be written in core notation or noble gas notation by replacing the1s2 2s2 2p6 3s. Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. Neon Ne Ne 3s 1.
The maximum number of electrons allowed in each of these subshells are 2 6 10 and 14 respectively. It indicates students forgot that K has one less electron than K. Its electron configuration is.
Its electron configuration will be Li. In order to write the I electron configuration we first need to know the number of electrons for the Barium Ba atom. K-- 2 L-- 8 M-- 8 N 1 In detail.
1s2 2s2 2p6 3s2 3p6 Comment to Instructor. The K shell contains a 1s. Write the electron configuration for a neutral atom of xenon.
Electron configuration of Helium He 1s 2. The full electron configuration of potassium k is 1s22s22p63s23p64s1. A bromine atom has two electrons in its first energy level eight electrons in its second 18 electrons in its third and seven electrons in its fourth.
Full electron configuration of xenon. H 1s1 He 1s2 Li 1s2 2s1 Be 1s2 2s2 B 1s2 2s2 2p1 C 1s2 2s2 2p2 N 1s2 2s2 2p3 O 1s2 2s2 2p4 F 1s2 2s2 2p5. 119 rows This decides the electron capacity of the shells.
There are 56 electrons for the Barium. 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6. Full Electron Configuration Electron shell arrangement.
Magnesium Mg Ne 3s 2 3p 1. Electron configuration can be described as how electrons are electron configuration long form. Hydrogen H 1s 2.
For example the names of the subshells in a sulfur atom would be 1s 2s 2p 3s and 3p since sulfur has three electron shells. The electron configuration for the first 10 elements. The electron configuration for Uranium U is based upon the placement of uranium in the fourth column of the actinide series or f block.
Rn 5f 14 6d 10 7s 2 7p 6. 1s 2 2s 1. Choice 2 indicates students added an extra electron.
Here is an example of what I mean. How to Write the Electron Configuration for Potassium K In order to write the Potassium electron configuration we first need to know the number of electrons for the K atom there are 19 electrons. Answer 1 of 10.
In the case of Krypton the abbreviated electron configuration is Ar 3d10 4s2 4p6. Alternatively write the symbol for the noble gas before an element radon in this case and just add the extra information. Iodine xenon cesium.
Keep in mind electron configurations are most stable when they are filled or half-filled. The electron configuration indicates that bromine has a total of 35 electrons. 1s 2 2s 2 2p 1.
For each atom the subshells are given first in concise form then with all subshells written out followed by the number of electrons per shell. Sodium Na Ne 3s 2. 1s2 2s2 2p6 3s2 3p6 4s1 2.
Aluminium Al Ne 3s 2 3p 2. Electron configuration of Lithium Li He 2s 1. Some are hard to memorise or predict so what is the electron configuration of an atom of Rb.
Well also look at why Lithium forms a 1 ion and how the electron configura. 1s 2 2s 2. 1s2 2s2 2p6 3s2 3p6 4s1 For more detail electronic configuration look at the image given below.
Atomic Symbol Electronic Configuration 19 K Ar 4s1 20 Ca Ar 4s2 21 Sc Ar 4s2 3d1 22 Ti Ar 4s2 3d2 23 V Ar 4s2 3d3 24 Cr Ar 4s2 3d4 25 Mn Ar 4s2 3d5 26 Fe Ar 4s2 3d6 27 Co Ar 4s2 3d7 28 Ni Ar 4s2 3d8 29 Cu Ar 4s2 3d9 30. Boron B He 2s 2 2p 2. Note that this is not always the same way they were added.
Some are hard to memorise or predict so what is the electron configuration of an atom of Kr.

What Is Electron Configuration For Cu 2

Beryllium Electron Configuration Electron Configuration Math Addition Worksheets Complex Sentences Worksheets
What Is The Electron Configuration For K Quora
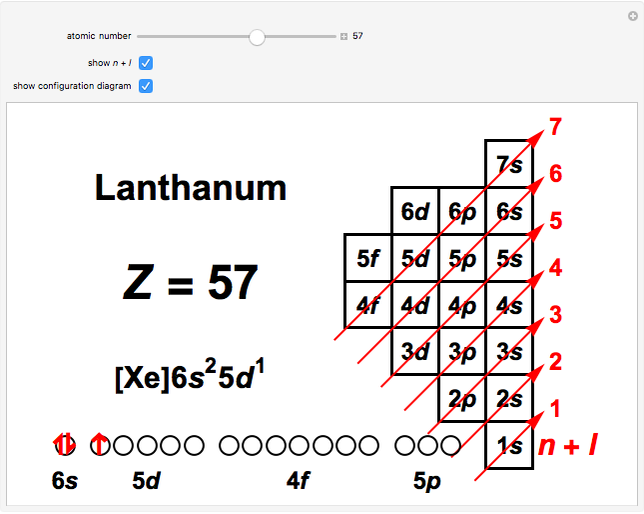
N L Rule For Atomic Electron Configurations Wolfram Demonstrations Project

5 Steps Electron Configuration For Phosphorus P In Just 5 Steps

Atom Diagrams Silicon Atom Electron Configuration Atom Diagram Atomic Structure

Electron Configuration Chemistry Anatomy And Physiology

Webelements Periodic Table Caesium Properties Of Free Atoms

File Electron Shell 019 Potassium Svg Wikimedia Commons Hyperkalemia Electron Configuration Nurse Rock

Chemistry291 Hand Note 5 Steps Electron Configuration For Potassium Or O In 2021 Electron Configuration Chemistry Lessons Configuration
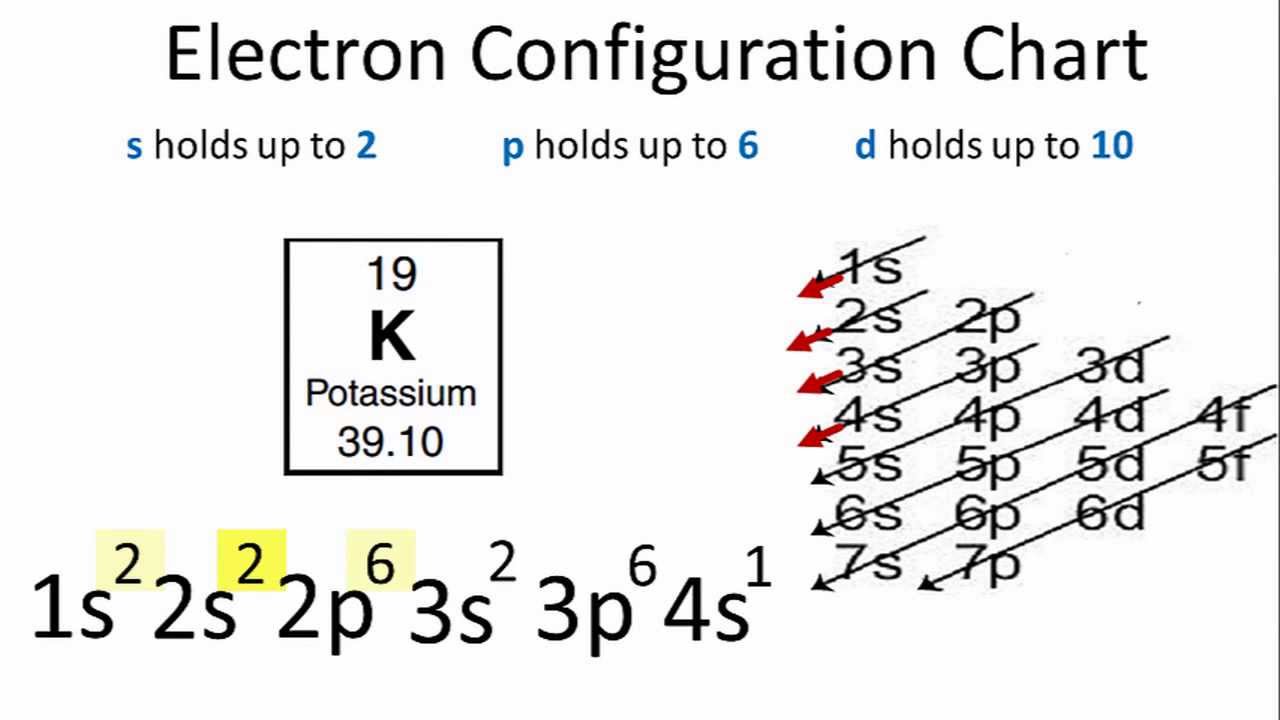
Electron Configuration For Potassium K

The Electron Configuration Of Potassium And Its Atomic Number Atom Model Bohr Model Atom Model Project
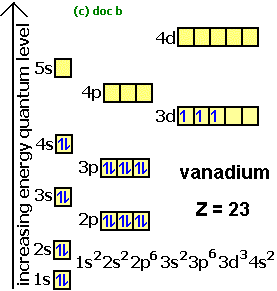
Electron Configurations How To Write Out The S P D F Electronic Arrangements Of Atoms Ions Periodic Table Oxidation States Using Orbital Notation Gce A Level Revision Notes

K Electron Configuration Potassium Ion Electron Configuration Electrons Configuration
Posting Komentar untuk "K Electron Configuration Long Form"