K+ Electron Configuration Noble Gas
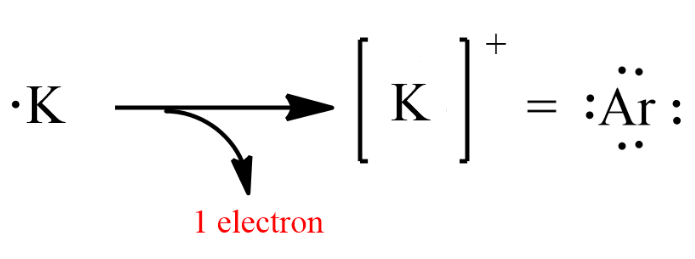
For example the K ion is isoelectronic with argon. Hence K is larger than K.
What Is The Formula Of The Ion Formed When Clutch Prep
The K ion has its outermost electrons on the third energy level now and is size is smaller than that of K ion.

K+ electron configuration noble gas. And finally we get. For an atom of potassium to attain octet configuration or noble gases configuration it will have to loose one electron thus forming a positively charged ion of potassium K. Express your answer as an integer.
A K b He c H- d O- e F- f Ca2 a Argon b no c Helium d no e Neon f Argon. This is favorable because it gives potassium a noble gas configuration. Indicate the number of unpaired electrons for following.
This provides the basis for a shorthand notation for electron configurations called the noble gas. Why is K larger than Ca2. E F- The atomic number of Fluorine is 9 so its atom has 9 electrons.
A phosphorus atom needs to gain __ electrons to achieve a noble gas configuration K Which of the following ions has the same electron configuration as an argon atom. Well also look at why Potassium forms a 1 ion and how the electron config. The Fluorine Ion F- has one negative charge so it will have 1 electron more which has negative charge thereby Fluorine ion.
When at neutral the potassium ion in the form of K has an electronic configuration of. Potassium K atom has 19 electrons. If there is more than one answer separate them by a comma.
Therefore electronic configuration of K and Ar will be same. K 19 refers to Potassium on the periodic table found in the Alkalai Metals first column on the left IA and in the fourth energy level row. A K d O-b He e F-c H-f Ca2.
The noble gas electronconfiguration is Ar4s1. What elements lack a noble gas electron configuration. For ionic bonding to occur metal atom must be bonded to a non-metal atom.
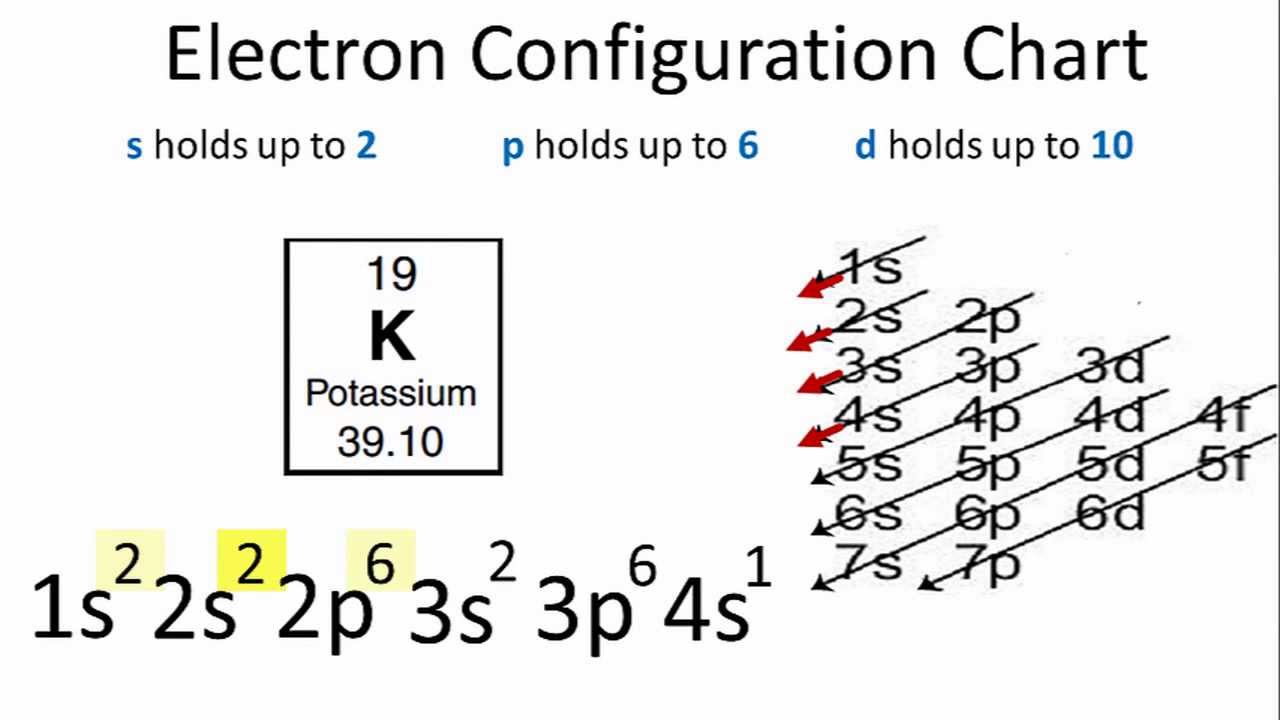
1s 2 2s 2 2p 6. The full electron configuration of Potassium K is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 1. For such configuration to happen in your syllabus when the question mentions LOSING GAINING electrons it is referring to ionic bonding.
1s22s22p63s23p6 configuration of argon Z18 4s1. What is the formula of the ion formed when sodium achieves noble gas electron configuration. Which of the following ions possess a noble gas electron configuration.
To achieve a noble gas electronic configuration the overall charge has to be 0 as noble suggest stable. Potassium has nineteen electrons one more than the noble gas argon so its configuration could be written as Ar4s1. Express your answer as the element symbol.
We talked about the fact that ions form because they can become more stable with the gain or loss of electrons to become like the noble gases and now. Its electron configuration is 1 s2 2 s2 2 p6 3 s1. K Electron Configuration Potassium Ion Electron configuration Electrons Configuration.
K GROUND STATE ELECTRON CONFIGURATION. Which of the following ions possess a noble gas electron configuration. Furthermore What is the formula of the ion formed when tin achieves a stable electron config 10th Chemistry What is the formula of the ion.
So we can replace this portion of potassiums electron configuration with the noble gas notation Ar I hope this was helpful. But we can abridge this slightly. Identify the elements that correspond to the following generalized electron configuration.
Here in this problem dont get confused with inert gas configuration and pseudo inert gas configuration. Potassium has 19 electrons and 1 is in the outermost shell. What is the formula of the ion formed when potassium achieves stability For an atom of potassium to attain octet configuration or noble gases configuration it will have to loose one electron thus forming a positively charged ion of potassium K.
B List the ions in order of increasing size. The first ten electrons of the sodium atom are the inner-shell electrons and the configuration of just those ten electrons is exactly the same as the configuration of the element neon Z 10. For example for Potassium K atomic 19 the preceding noble gas is Argon Ar atomic 18.
In case of pseudo inert configuration there should be 18 electrons in the outermost shell. There is a large increase in the second ionization energy for K compared to Ca because removal of the second electron from K is a core electron that is in a. Oct 29 2019 - In this video we will write the electron configuration for K the Potassium ion.
Therefore K ion possess a noble gas electon configuration. So only Cuhas pseudo inert gas configuration and all others have inert gas configuration. With 10 electrons you should note that oxygens electron configuration is now exactly the same as Neons.
Potassium ion K has the same number of electrons 18 as noble gas Argon. Does K have a noble gas electron configuration. The first ionization energy for K is less than Ca because Ca has a larger effective nuclear charge.
Potassium K has an atomic number of 19. Thus substituting the config of He gives the full config for Neon. For potassium Z 19 and so for the full electronic configuration we write.
Is K isoelectronic with a noble gas. Find the electron configuration for Potassium K - YouTube. So the correct answer is B.
If potassium is ionized and becomes a 1 cation this is to say that it lost its single valence electron. Potassium is the FIRST element in the FOURTH row of the tableSo its the same as Argon but with one extra electron in its 4s subshell. 119 rows ELECTRON CONFIGURATION.
Thus the configuration shown for Potassium is Ar4s 1 see Table below. Here only Cu has 18 electrons in its outer shell. K has four shells S only has three.
Energy level Wikipedia K Potassium Electron Configuration Noble Gas Notation IB Chemistry on Quantum Numbers and Electronic Configuration Schalenmodell Kernphysik â Wikipedia Shorthand Electron Configuration and Valence Electrons The Atom.

Potassium Electron Configuration K With Orbital Diagram

K Electron Configuration Potassium Ion Electron Configuration Electrons Configuration

Noble Gas Configuration Shorthand Electron Configuration
What Is An Isoelectronic Series Quora

Potassium Electron Configuration K With Orbital Diagram

Ch 6 Review Periodic Table Do You Have

Potassium Electron Configuration K With Orbital Diagram
Potassium Electron Configuration K With Orbital Diagram

K Electron Configuration Potassium Ion Youtube
What Is The Electron Configuration For K Quora

Find The Electron Configuration For Potassium K Youtube

Question Video Identifying The Condensed Electronic Configuration Of A Potassium Atom Nagwa

How Many Valence Electrons Are In An Atom Of Magnesium Ppt Video Online Download

Posting Komentar untuk "K+ Electron Configuration Noble Gas"